 With this simple trend in mind, we can decode all other period trends. The elements on the Periodic Table are arranged in order of increasing. On the other hand, as we go down a column (called a group), we are adding energy levels, meaning that our electrons are located further away from the nucleus and thus, by Coulomb’s law, the effective nuclear charge force felt by valence electrons is less. This means that the effective nuclear charge felt by valence electrons is greater. Also, one more thing to memorize on the periodic table, is periodic trends. 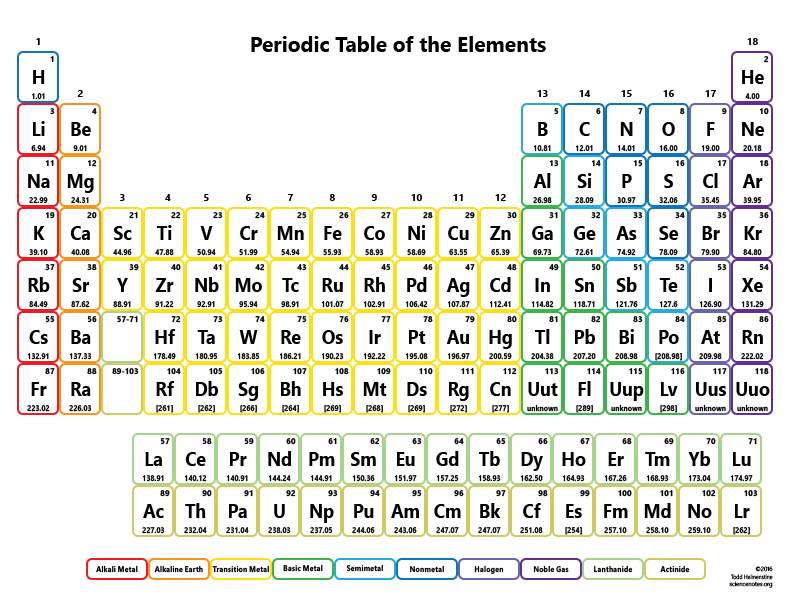
The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. This is because we are adding protons without adding any new energy levels. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. As we go down a row (or period) in the periodic table, the effective nuclear charge increases. Valence electrons are simply the electrons in the most outer energy level. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Effective nuclear charge is a measure of the average nuclear charge felt by valence electrons given shielding from the inner-shell electrons. However, there is a simple trick that explains all periodic trends based solely on effective nuclear charge. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. 
If you’ve taken a chemistry class before, you likely learned about periodic trends, which can be cumbersome to remember. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
